Ipratropium bromide formulations
Ipratropium bromide has an anticholinergic effect
Ipratropium bromide has an anticholinergic effect, blocking the action of acetylcholine on muscarinic receptors, leading to a decrease in watery nasal secretions (runny nose) and sneezing.2It has a strong synergistic effect with xylometazoline.1
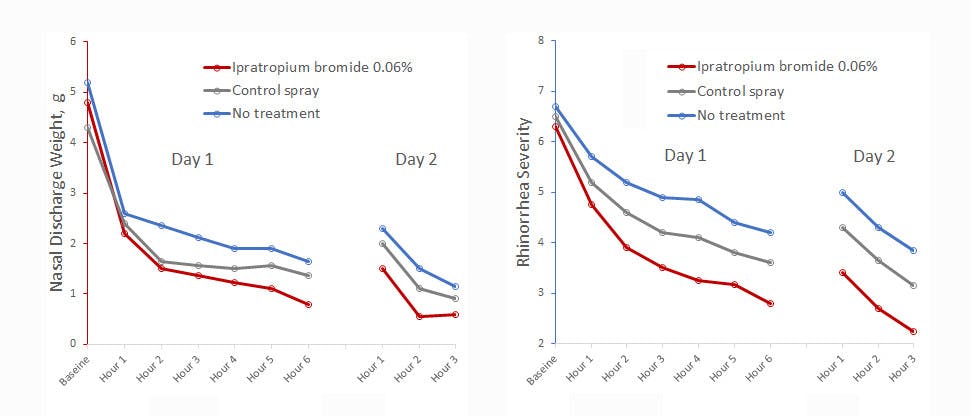
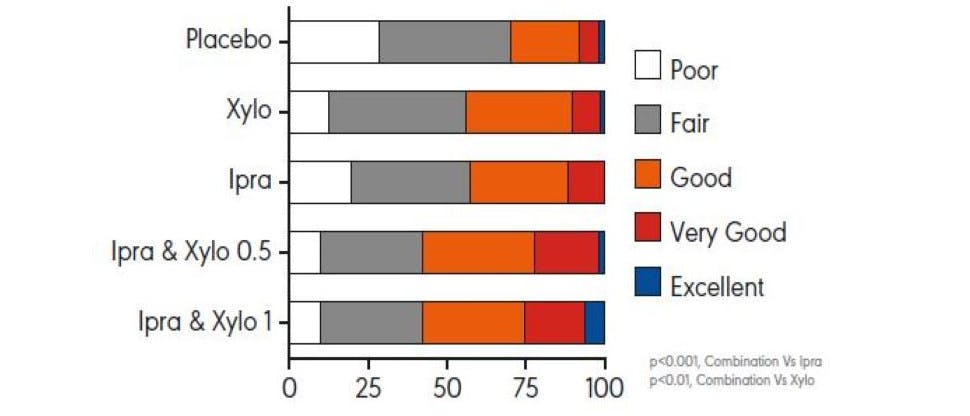
A clinical study demonstrates an improvement in the severity of nasal discharge with ipratropium bromide nasal spray3
Patients treated with ipratropium bromide nasal sprays demonstrated 26% less nasal discharge compared to controls treated with a buffered salt solution nasal spray, and 34% less discharge compared to untreated patients.3 In the same study, the severity of nasal discharge was also subjectively judged to be reduced by 78% in participants taking ipratropium nasal spray when compared to untreated participants.3
Ipratropium bromide reduces the symptoms of nose dripping and sniffing, often present with the common cold4,5
Discover the Otrivin range with ipratropium bromide

Otrivin Dual Relief
Otrivin Dual Relief unblocks the nose and reduces runniness with ipratropium bromide.
Otrivin – your Nasal HealthPartner